We are on track for IDE Submission to use RadioGel™ in human clinical trials.
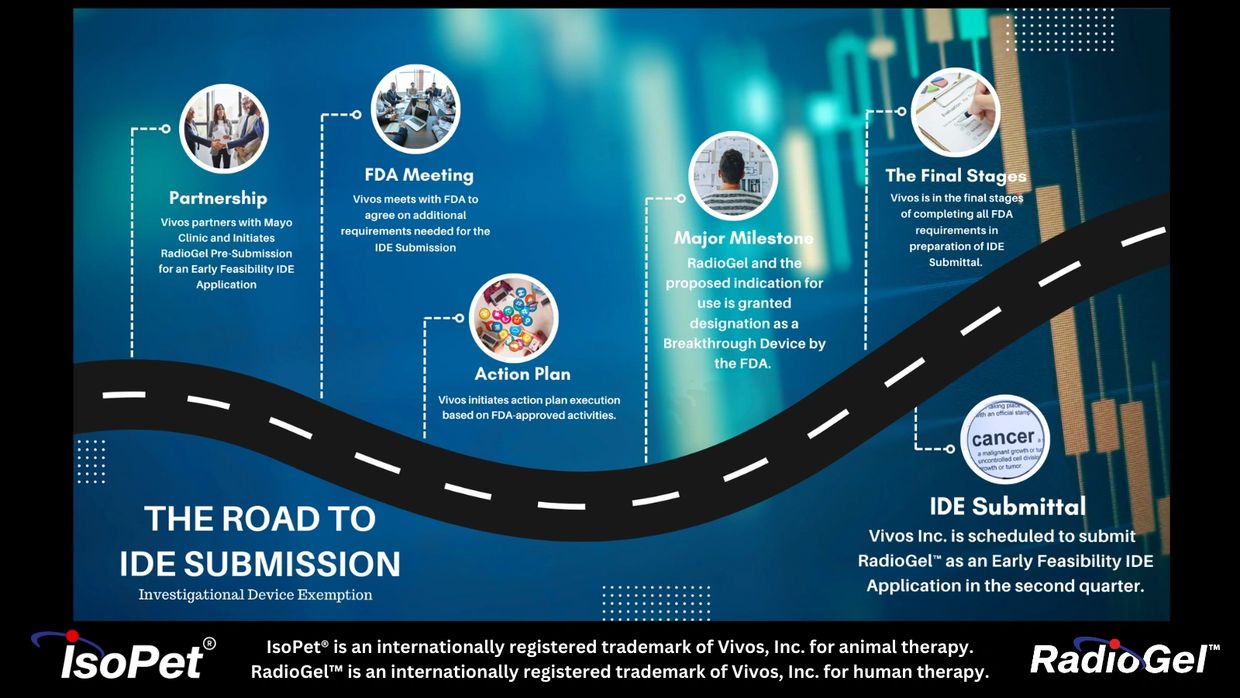
IDE Submission
A New Generation Cancer Treatment
Our RadioGel™ division assembled a team of talented and dedicated professionals who are passionate about improving cancer treatment through cutting edge technology and an advance knowledge for innovative solutions to cancer therapy. Our team includes brilliant scientists, dedicated researchers, innovative engineers, and top notch business professionals, all of whom bring unique skills and perspectives to our work.
We are in the final stages of preparing to submit the necessary documents for IDE Submission.
RadioGel™ is currently not approved for human use.
Learn More
Investigations covered under the IDE regulation are subject to differing levels of regulatory control depending upon the level of risk. The IDE regulation distinguishes between significant and nonsignificant risk device studies and the procedures for obtaining approval to begin the study differ accordingly. Also, some types of studies are exempt from the IDE regulations.
Updates
Mayo Clinic Update
Johns Hopkins Update
Johns Hopkins Update

Johns Hopkins Update
Johns Hopkins Update
Johns Hopkins Update
